Abstract
Background: CAR (chimeric antigen receptor) T-cell therapy is a novel form of immunotherapy that utilizes genetically altered autologous T cells to target cancer cells. Since the first FDA (Food and Drug Administration) approval in August 2017, several CAR T-cell products have been approved for the treatment of various malignancies. Due to the recent approval, large-scale epidemiologic data are lacking. This study aims to characterize the epidemiology of hospitalizations for CAR T-cell therapy, readmissions, and factors associated with all-cause-30-day readmission.
Methods: We performed a retrospective cohort study using the Nationwide Readmissions Database (NRD) for 2017 & 2018 to identify hospitalizations with CAR T-cell therapy administration utilizing the ICD-10-PCS (International Classification of Diseases, Tenth Revision, Procedure Coding System) codes XW033C3 & XW043C3. These admissions were further categorized into non-Hodgkin's lymphoma, leukemia, and multiple myeloma. Descriptive analysis was performed to compare demographics, hospital characteristics, various comorbid conditions, and complications between these groups. We identified primary diagnoses at readmission and all-cause 30-day readmission rate after excluding index admissions from December and deaths during index admissions. Multivariable logistic regression was used to elucidate factors associated with all-cause-30-day readmission. The analysis was carried out to produce national estimates after applying weights, and the methodology provided by the HCUP (Healthcare Cost and Utilization Project) was utilized.
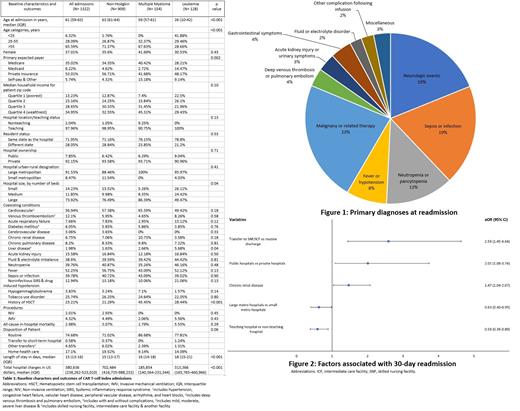
Results: Out of 1,322 CAR T-cell therapy admissions in 2017-2018, 909 had a diagnosis of non-Hodgkin's lymphoma, 154 had multiple myeloma, and 128 had leukemia. The median age of those with leukemia was lowest at 26 (10-42) years, followed by multiple myeloma and non-Hodgkin's at 59 (57-61) years and 63 (61-64) years, respectively (p<0.001). The majority of CAR T-cell recipients were males (63%), admitted to teaching hospitals (97.96%), had private insurance (53.01%), and belonged to the highest income communities (34.95%). The median length of stay was longest for leukemia, followed by multiple myeloma and non-Hodgkin's (18 days vs. 16 days vs. 15 days; p<0.001). Median hospital charges were highest for non-Hodgkin's, followed by leukemia and multiple myeloma ($702,484 vs. $313,366 vs. $185,854; p<0.001). Overall mortality during index hospitalization was 2.98%. The complete baseline characters are described in table 1. A total of 316 hospitalizations (25%) were readmitted within 30 days. The median time to readmission was 9 days, the median length of stay during readmission was 5 days, and crude in-hospital mortality during readmission was 3%. Readmission incurred an additional median hospital charge of $58,568. The top five diagnoses at readmission were malignancy & therapy-related (23%), sepsis or infection (19%), neurologic events (19%), neutropenia or pancytopenia (13%), and fever or hypotension (8%) (figure1). On multivariable logistic regression for predictors of 30-day readmission, admission to public hospital (aOR {adjusted odds ratio} 2.01, p 0.027), transfer to a skilled nursing facility or intermediate care facility at discharge (aOR 2.59, p 0.001), and chronic renal disease (aOR 1.47 p 0.027) were associated with higher odds of readmission. Admission to a large metro hospital (aOR 0.63 p 0.047) and teaching hospital (aOR 0.59 p 0.012) were associated with reduced odds of readmission (figure2).
Conclusion: As CAR T-cell therapy has recently been added to the cancer treatment arsenal, we describe that it is associated with strikingly high hospital charges, and a quarter of recipients are readmitted within the first 30 days.
Deol: Kite, a Gilead Company: Consultancy.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal